Liquid-Liquid Extraction (LLE) is a mass transfer operation that is used to separate components of a liquid mixture by contacting it with another liquid. It must have at least two components. Usually, we are interested in separating only one component from a feed stream. This component is called a ‘Solute’. The components other than the Solute are together referred to as carrier liquid. The Solvent stream is usually a pure liquid.
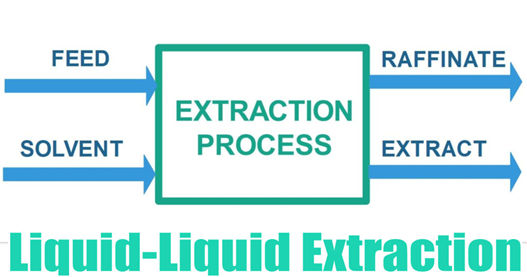
The surface tension of the liquids should be such that it must keep them separated after the mixing is done and not prohibit the movement of tiny droplets into the other phase during the mixing process itself. The tinier the droplets, the faster is the mass transfer rate of the solute from the feed to the solvent. If the density difference of both the liquids is not large enough, then after the mixing is done, both the phases won’t separate easily or instantly and will exist as dispersion. This dispersion is called an Emulsion.
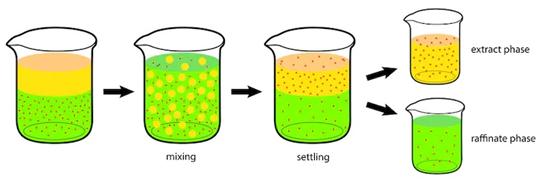
The Emulsion can be broken by working with parameters that affect liquid density, such as increasing the Extraction Process Feed Solvent Raffinate Extract temperature of the process or adding another component that helps in the separation of the phases. After the mixing process is done, it separates into two phases, the solute rich phase is called an Extract phase and ideally it is made up of Solute and Solvent. The solute lean phase is called Raffinate phase, it is mostly made up of little bit of solute and the carrier liquid.
Extraction may be a process of choice when:
- Distillation cannot be used if the components to be separated have close boiling point temperatures or the components to be separated are sensitive to heat.
- The amount of solute is very less in the feed itself, in essence a dilute liquid mixture.
- We have to separate non-volatile metal solutes in metal extraction industries.
- We have to remove organics from the aqueous stream
- Source : chemicalengineeringworld.com

No Responses